Greenlight Guru
Cloud-based electronic quality management system (eQMS) purpose-built for medical device companies. Manages design controls, risk management, CAPA, document control, and clinical data capture with built-in FDA 21 CFR Part 11 compliance.
Greenlight Guru is a cloud-based electronic quality management system (eQMS) designed specifically for medical device manufacturers. The platform provides an integrated suite for managing design controls, risk management, quality processes, and clinical data capture throughout the entire product lifecycle.
Core capabilities
Design and development
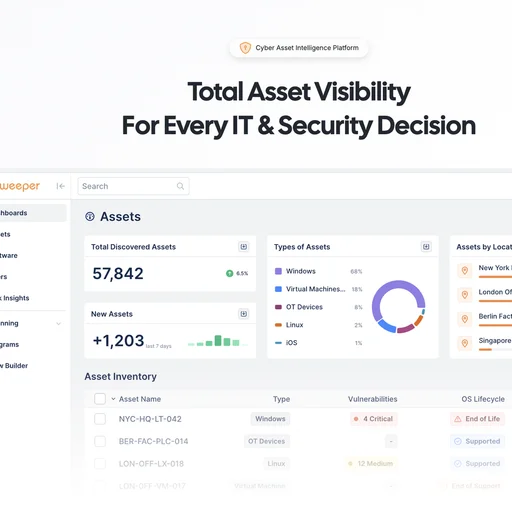
- Digital Design History File (DHF) management with built-in traceability
- Link design inputs, outputs, verifications, and product risk controls
- AI-powered suggestions and predictive verifiability checks
- Design controls module with cross-functional collaboration features
Compliance and risk management
- Operational risk tracking across the QMS
- Regulatory requirements management
- Audit readiness tools and CAPA trend analysis
- Built-in compliance with FDA 21 CFR Part 11
Documents and training management
- Centralized document control with automated routing
- Audit trails and version control
- Role-based access control
- Training management and competency tracking
Quality processes and events
- CAPA (Corrective and Preventive Action) management
- Non-conformance tracking
- Supplier quality management
- Connected system linking quality events to training and documentation
Clinical data management
- Electronic Data Capture (EDC) for clinical studies
- Audit-ready clinical data across all study phases
- First-in-human through post-market study support
Limitations
- SaaS-only deployment: No on-premise or self-hosted option available, requiring internet connectivity and reliance on vendor infrastructure
- Medical device focus: Purpose-built for MedTech; not suitable for general manufacturing quality management or non-regulated industries
- Custom pricing: No public pricing tiers; requires sales engagement for quotes, making cost evaluation difficult for small companies
- Integration constraints: Limited native integrations with ERP, PLM, or MES systems common in manufacturing environments
- Industry scope: While supporting FDA regulations, may require additional configuration for EU MDR or other international regulatory frameworks
Categories: